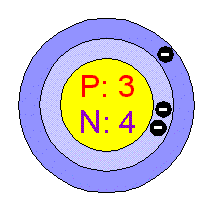
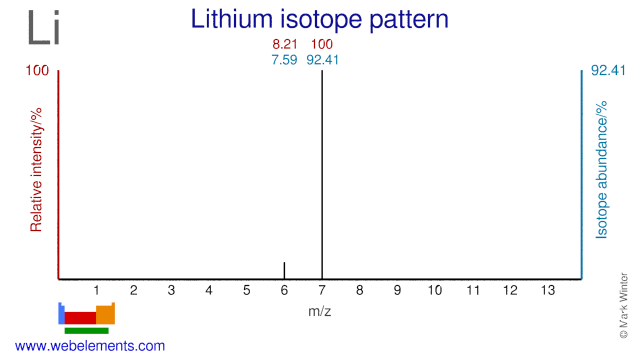
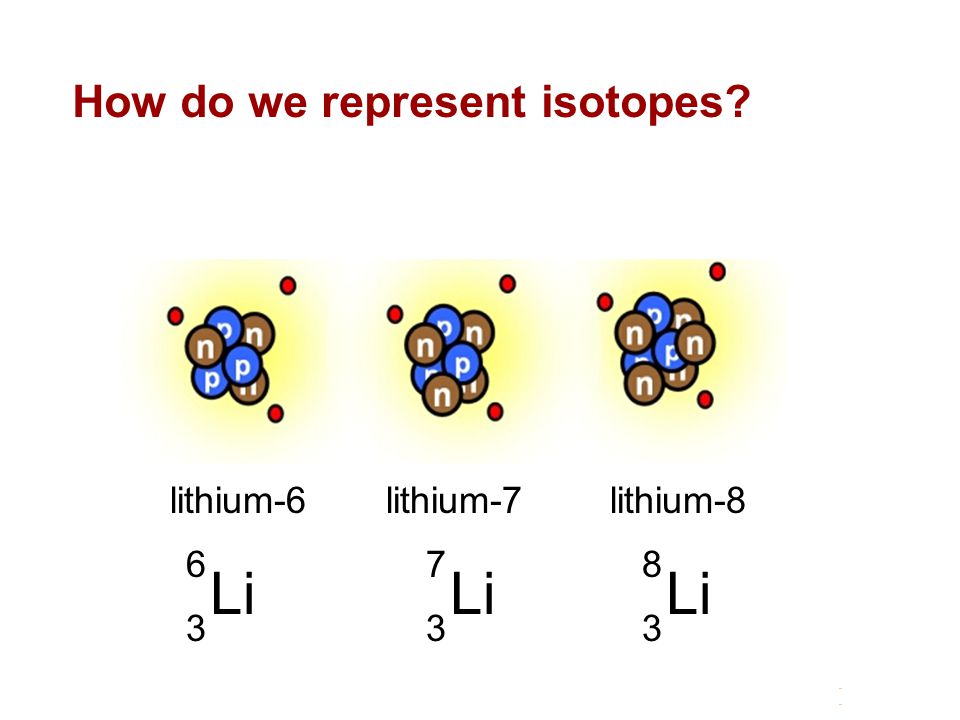
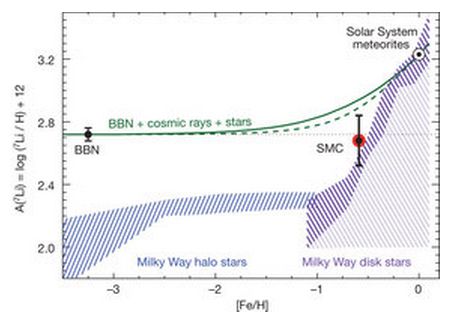
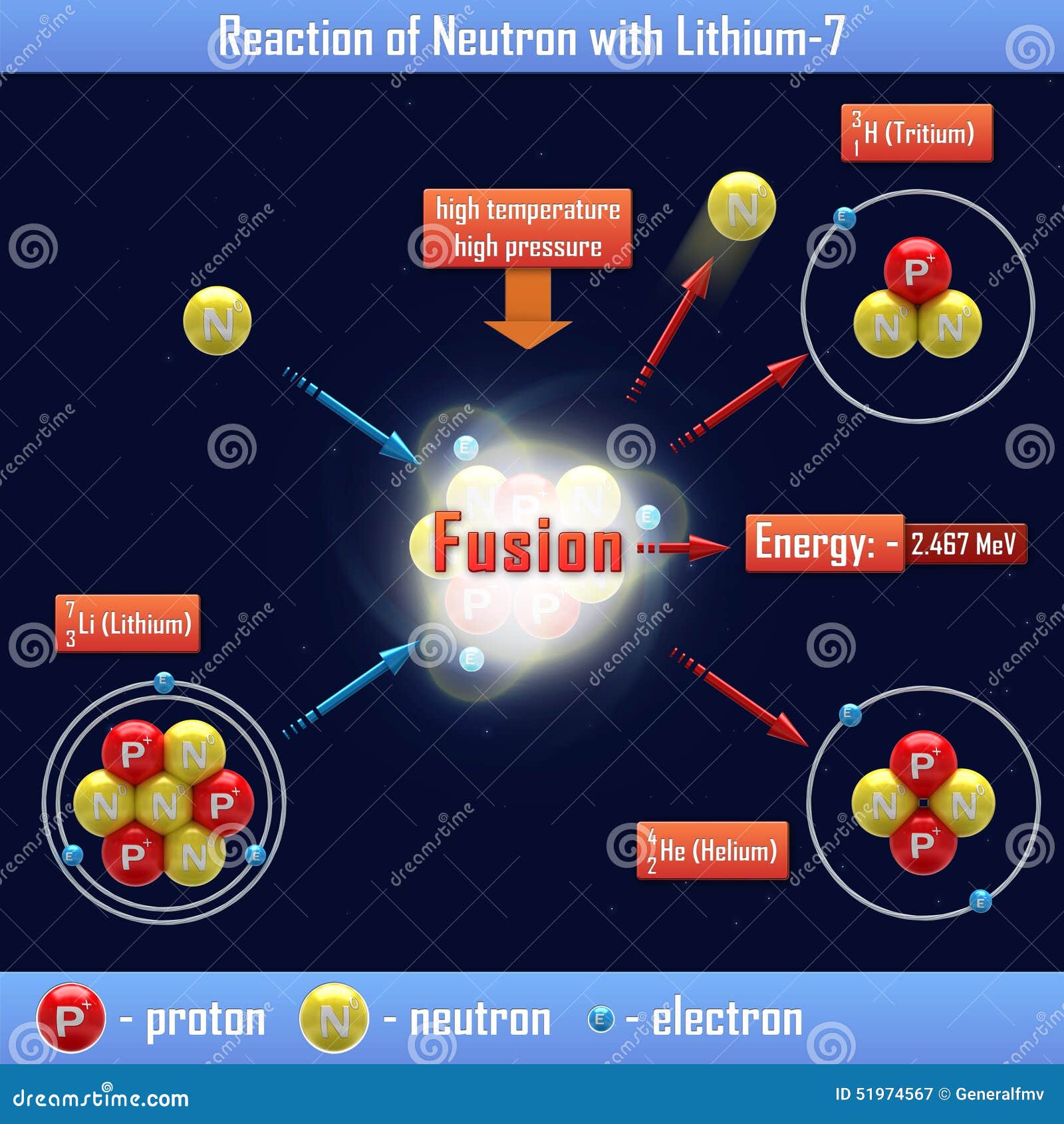
SOLVED: 9. Why are all the values in each row of the table above the same? 10. Lithium has only two stable isotopes. Use the sim to determine the following: a. Atomic

Determine the atomic mass of lithium, given its isotopic composition of 92.4% lithium-7 (mass 7.016 amu) and 7.60% lithium-6 (mass 6.015 amu). | Homework.Study.com